A) 1s22s22p2
B) 1s22s32p3
C) 1s22s32p2
D) 1s22s22p4
E) 1s22s22p3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Light of wavelength 411 nm is observed from a hydrogen discharge. What transition produces this emission? The energy of the n = 1 level is -13.6 eV.
A) n = 1.13 to n =1
B) n = 6 to n = 2
C) n = 3 to n = 3
D) n = 2 to n = 6
E) n = 5 to n = 1
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
J. J. Thomson's model of an atom
A) had electrons embedded in some kind of fluid that contained most of the mass of the atom.
B) held that the fluid containing most of the mass of the atom had enough positive charge to make the atom electrically neutral.
C) failed to predict the observed frequencies for any atom.
D) depended upon electric forces to produce stability.
E) All of these are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equation derived by Bohr for the wavelengths of of the lines in hydrogen- like spectra is 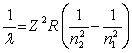 The first member of the Balmer series of hydrogen has = 660 nm. Doubly ionized
The first member of the Balmer series of hydrogen has = 660 nm. Doubly ionized 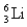 is hydrogen-like. The wavelength of the first member of the Balmer series for doubly ionized
is hydrogen-like. The wavelength of the first member of the Balmer series for doubly ionized 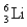 is
is
A) 73 nm
B) 5.9 * 103 nm
C) 150 nm
D) 60 nm
E) 1.8* 10-3 nm
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
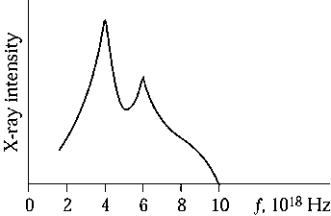 The graph shows the spectrum of X rays produced by the bombardment of a metal target with high-energy electrons. The maximum kinetic energy of the electrons striking the target is
The graph shows the spectrum of X rays produced by the bombardment of a metal target with high-energy electrons. The maximum kinetic energy of the electrons striking the target is
A) 13.3 * 10-16 J
B) 66.3 *10-16 J
C) 26.5 * 10-16 J
D) 39.8 * 10-16 J
E) 4 *10-18 J
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The orbital angular momentum L is related to the orbital quantum number 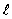 by
by
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following characteristic X-ray lines results from the least energetic transition?
A) K
B) K
C) K
D) All of these characteristic X-ray lines are the same.
E) None of these is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Mosely showed that the wavelength of K characteristic X-ray radiation is directly related to the nuclear property of
A) atomic number.
B) mass number.
C) neutron number.
D) mass defect.
E) binding energy per nucleon.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 128 of 128
Related Exams