B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The characteristic way in which atoms of an element react is most related to the
A) number of electrons in the outermost shell.
B) number of electrons in the innermost shell.
C) number of neutrons in the nucleus.
D) size of the nucleus.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Not Answered
Study the figures to determine which is liquid water and which is frozen water (ice).Explain your answer and predict if the water in Figure 2 would float or sink in the water in Figure 1. 
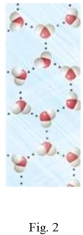
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is NOT one of the six most common elements in living organisms?
A) carbon
B) oxygen
C) iron
D) nitrogen
E) hydrogen
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electrons are unequally shared in _______,and transferred in __________.
A) CH4, Na+Cl-
B) O2,CH4
C) Na+Cl-,H2O
D) H2O,N2
F) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
A change of one pH unit represents a ten-fold increase or decrease in hydroxyl ion concentration.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
pH 7 has a balanced level of H+ and OH-.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a water molecule,
A) the oxygen atom is more electronegative than the hydrogen atoms.
B) the oxygen atom has an overall negative charge with the hydrogen atoms having an overall positive charge.
C) unequal sharing of electrons results in a polar molecule.
D) All of the choices are correct.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Not Answered
Classify the following substances as either hydrophobic or hydrophilic:

Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements would be more reactive with other elements?
A) boron,#5
B) neon,#10
C) argon,#18
D) helium,#2
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is NOT true about electron configurations?
A) If an atom has only one shell,it is complete with two electrons.
B) If an atom has two or more shells,the octet rule applies.
C) If an atom has two or more shells,the outer shell is complete with eight electrons.
D) Atoms with more than eight electrons in the outer shell react by gaining electrons.
E) Atoms with eight electrons in the outer shell are not reactive at all.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Not Answered
Use Bohr's model to draw a sodium (Na)atom and a chlorine (Cl)atom.Using your model,explain what happens when sodium reacts with chlorine to form table salt.Include in your explanation ion and ionic bond formation.Use your model to help you to decide whether NaCl is hydrophilic or hydrophobic. 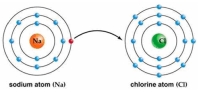
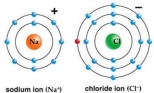
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A research article indicates that researchers have used an isotope 3H to trace a certain metabolic process.From the symbol that is given,we know this is a hydrogen isotope with
A) three protons.
B) three neutrons.
C) three electrons.
D) one proton and two neutrons.
E) two protons and one neutron.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Prior to prescription medications to control stomach acid and "heart burn" people consumed baking soda (sodium bicarbonate) to decrease their discomfort.This would indicate that sodium bicarbonate
A) effectively buffers stomach acid by releasing H+
B) should be sold as a prescription drug
C) blocks acid production by combining with OH-
D) neutralizes stomach acid by combining with excess H+
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As a solid,water floats.This means that
A) solid water is less dense than liquid water.
B) organisms in ponds,lakes,and reservoirs can survive under the ice cover.
C) this is due to hydrogen bonding changes.
D) All of the choices are correct.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which term refers to the attraction to water molecules?
A) hydrophilic
B) hydrophobic
C) hydrolysis
D) photolysis
E) nitrophylic
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The scale indicates the relative concentrations of hydrogen and hydroxyl ions in a solution.
B) False
Correct Answer

verified
Correct Answer
verified
Not Answered
Draw two hydrogen atoms using Bohr's model.Now bond them to form a molecule of hydrogen gas.Write the molecular formula.Explain what type of bond you've created and why this is a stable situation.
Correct Answer

verified
Correct Answer
verified
Not Answered
 Study the chart to determine the relationship between H+ concentration and pH.If you were to create a herbal remedy to decrease excess stomach acid,would you create a solution with a relatively greater or lesser number of hydrogen ions.
Study the chart to determine the relationship between H+ concentration and pH.If you were to create a herbal remedy to decrease excess stomach acid,would you create a solution with a relatively greater or lesser number of hydrogen ions.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Human blood has a pH of about 7.4.This is
A) neutral.
B) very acidic.
C) slightly acidic.
D) slightly basic.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 57
Related Exams