B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
What is the mole fraction of Ne in a mixture containing 10.1 g of Ne, 79.9 g of Ar, and 83.8 g of Kr?
A) 0.40
B) 0.25
C) 0.20
D) 0.14
E) 0.058
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Procaine hydrochloride (  = 272.77 g/mol) is used as a local anesthetic. Calculate the molarity of a 4.666 m solution which has a density of 1.1066 g/mL.
= 272.77 g/mol) is used as a local anesthetic. Calculate the molarity of a 4.666 m solution which has a density of 1.1066 g/mL.
A) 2.272 M
B) 4.056 M
C) 4.216 M
D) 4.666 M
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
A saturated solution of carbon dioxide in water contains 3.00 g of CO2 when the CO2 partial pressure is 8.0 atm. What mass of CO2 will escape if the partial pressure is lowered to 3.2 atm?
A) 0.90 g
B) 1.20 g
C) 1.40 g
D) 1.80 g
E) 2.20 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Electrolyte solutions generally behave less ideally as the solute concentration increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a given solution, which of the following concentration values will change as temperature changes?
A) mass percent
B) molality
C) mole fraction
D) molarity
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following sets of conditions could exist when two liquids which are completely miscible in one another are mixed?
A) ΔHsoln > 0, entropy of system decreases
B) ΔHsoln ≈ 0, entropy of system decreases
C) ΔHsoln ≈ 0, entropy change of system ≈ 0
D) ΔHsoln ≈ 0, entropy of system increases
E) None of these choices are correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The vapor pressure of pure acetone (propanone) is 266 torr. When a non-volatile solute is added, the vapor pressure of acetone above the solution falls to 232 torr. What is the mole fraction of acetone in the solution?
A) 0.87
B) 0.69
C) 0.32
D) 0.13
E) 0.045
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Soda drinks bubble when the bottle is opened because
A) the temperature of the soda increases.
B) exposure to atmospheric pressure squeezes the carbon dioxide from solution.
C) the partial pressure of carbon dioxide above the solution is reduced.
D) atmospheric nitrogen molecules displace carbon dioxide molecules.
E) the Henry's law constant changes due to the change in pressure.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements describes the correct method of preparation of 1.00 L of a 2.0 M urea solution? 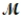 urea = 60.06 g/mol
urea = 60.06 g/mol
A) Dissolve 120 g of urea in 1.00 kg of distilled water.
B) Dissolve 120 g of urea in 880 g of distilled water.
C) Dissolve 120 g of urea in enough distilled water to produce 1.00 L of solution.
D) Dissolve 120 g of urea in 1.00 liter of distilled water.
E) The density of urea is needed in order to do this calculation.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions will be expected to have the most negative heat of hydration, ΔHhydr?
A) Na+
B) Cs+
C) Ca2+
D) F-
E) I-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Tyndall effect
A) is observed in concentrated solutions.
B) is observed only in dilute solutions.
C) is observed in colloidal dispersions.
D) is caused by Brownian motion.
E) is used to determine the osmotic pressure of solutions.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pairs of dispersed phases and dispersing media can never form a colloid?
A) solid and gas
B) liquid and gas
C) solid and solid
D) liquid and liquid
E) gas and gas
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The shape of a protein molecule is determined completely by
A) hydrogen bonding.
B) ion-dipole attractions.
C) dispersion forces.
D) disulfide bridges.
E) the sequence of amino acids in the chain.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 2.0% (w/v) solution of sodium hydrogen citrate, Na2C6H6O7, which also contains 2.5% (w/v) of dextrose, C6H12O6, is used as an anticoagulant for blood which is to be used for transfusions. What is the molarity of the sodium hydrogen citrate in the solution?
A) 0.085 M
B) 0.19 M
C) 0.53 M
D) 1.2 M
E) 1.3 M
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Dimethylglyoxime, DMG, is an organic compound used to test for aqueous nickel(II) ions. A solution prepared by dissolving 65.0 g of DMG in 375 g of ethanol boils at 80.3°C. What is the molar mass of DMG? Kb = 1.22°C/m, boiling point of pure ethanol = 78.5°C
A) 44.1 g/mol
B) 65.8 g/mol
C) 117 g/mol
D) 131.6 g/mol
E) 553 g/mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which, if any, of the following features is common to soaps, detergents, phospholipids, and channel-forming antibiotics?
A) They all contain fatty acids.
B) They all contain phosphate groups.
C) They all contain polypeptide chains.
D) Their function depends on the dual polarity of their molecules.
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What concentration of aqueous FeCl3 would have the same osmotic pressure as a 0.20 M solution of CaCl2 at the same temperature, assuming ideal behavior?
A) 0.60 M
B) 0.40 M
C) 0.30 M
D) 0.15 M
E) 0.10 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two aqueous are prepared: 1.00 m Na2CO3 and 1.00 m LiCl. Which of the following statements is true?
A) The Na2CO3 solution has a higher osmotic pressure and higher vapor pressure than the LiCl solution.
B) The Na2CO3 solution has a higher osmotic pressure and higher boiling point than the LiCl solution.
C) The Na2CO3 solution has a lower osmotic pressure and lower vapor pressure than the LiCI solution.
D) The Na2CO3 solution has a lower osmotic pressure and higher boiling point than the LiCl solution.
E) None of these choices are correct.
G) B) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
How many moles of sulfate ions are present in 1.0 L of 0.5 M Li2SO4?
A) 0.5 mol
B) 1.0 mol
C) 1.5 mol
D) 2.0 mol
E) 3.0 mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 96
Related Exams