A) ΔH°f = 0 for Al(s) at 298 K
B) ΔG°f = 0 for H2(g) at 298 K
C) S° = 51.446 J/(mol·K) for Na(s) at 298 K
D) qsys < 0 for H2O(l) → H2O(s) at 0°C
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which, if any, of the following processes is spontaneous under the specified conditions?
A) H2O(l) → H2O(s) at 25°C
B) CO2(s) → CO2(g) at 0°C
C) 2H2O(g) → 2H2(g) + O2(g)
D) C(graphite) → C(diamond) at 25°C and 1 atm pressure
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elemental boron can be formed by reaction of boron trichloride with hydrogen. BCl3(g) + 1.5H2(g) → B(s) + 3HCl(g) Calculate ΔG° for the reaction. Substance: BCl3(g) H2(g) B(s) HCl(g) ΔG°f (kJ/mol) : -388.7 0 0 -95.3
A) -293.4 kJ
B) 293.4 kJ
C) -102.8 kJ
D) 102.8 kJ
E) None of these choices are correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the figure that shows ΔG° for a chemical process plotted against absolute temperature. 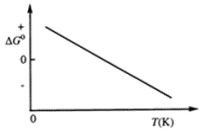 Which one of the following is an incorrect conclusion, based on the information in the diagram?
Which one of the following is an incorrect conclusion, based on the information in the diagram?
A) ΔH° > 0
B) ΔS° > 0
C) The reaction is spontaneous at high temperatures.
D) ΔS° increases with temperature while ΔH° remains constant.
E) There exists a certain temperature at which ΔH° = TΔS°.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true for a system at equilibrium?
A) ΔS°sys = ΔS°surr
B) ΔS°sys = -ΔS°surr
C) ΔS°sys = ΔS°surr = 0
D) ΔS°univ > 0
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
For any reaction, if ΔG° > 0, then K < 1.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs has the member with the greater molar entropy listed first? All systems are at 25°C.
A) CO(g) , CO2(g)
B) NaCl(s) , NaCl(aq)
C) H2S(g) , H2S(aq)
D) Li(s) , Pb(s)
E) H2(g) , H2O(g)
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The free energy of a perfect crystal at absolute zero, is zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? 2H2S(g) + 3O2(g) → 2H2O(g) + 2SO2(g)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a chemical reaction to be spontaneous at all temperatures, which of the following conditions must be met?
A) ΔS° > 0, ΔH° > 0
B) ΔS° > 0, ΔH° < 0
C) ΔS° < 0, ΔH° < 0
D) ΔS° < 0, ΔH° > 0
E) It is not possible for a reaction to be spontaneous at all temperatures.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen sulfide decomposes according to the following reaction 2H2S(g) → 2H2(g) + S2(g) For this reaction at 298K ΔS° = 78.1 J/K, ΔH° = 169.4 kJ, and ΔG° = 146.1 kJ. What is the value of ΔG° at 900 K?
A) -69881 kJ
B) 48.4 kJ
C) 99.1 kJ
D) 240 kJ
E) 441 kJ
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction CuI(s)  Cu+(aq) + I-(aq)
If the concentrations of the Cu+ and I- ions in equilibrium at 298 K are both equal to 1.03 × 10-6 M, what is the value of ΔG° for the reaction?
Cu+(aq) + I-(aq)
If the concentrations of the Cu+ and I- ions in equilibrium at 298 K are both equal to 1.03 × 10-6 M, what is the value of ΔG° for the reaction?
A) -68 kJ
B) 68 kJ
C) -30. kJ
D) 30 kJ
E) 34 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship best describes ΔS° for the following reaction? 8H2(g) + S8(s) → 8H2S(g)
A) ΔS° = ΔH°
B) ΔS° = ΔH°/T
C) ΔS° ≈ 0
D) ΔS° < 0
E) ΔS° > 0
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the equilibrium constant at 25°C for the reaction of methane with water to form carbon dioxide and hydrogen. The data refer to 25°C. CH4(g) + 2H2O(g)  CO2(g) + 4H2(g)
CO2(g) + 4H2(g)
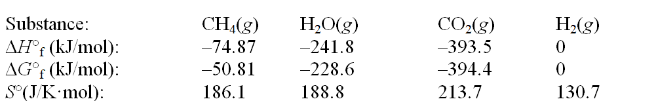
A) 8.2 × 1019
B) 0.96
C) 0.58
D) 1.2 × 10-20
E) 1.4 × 10-46
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide. H2O2(l)  H2O2(g)
Use the following thermodynamic information at 298 K to determine this temperature.
Substance: H2O2(l) H2O2(g)
ΔH°f (kJ/mol) : -187.7 -136.3
ΔG°f (kJ/mol) : -120.4 -105.6
S°(J/K·mol) : 109.6 232.7
H2O2(g)
Use the following thermodynamic information at 298 K to determine this temperature.
Substance: H2O2(l) H2O2(g)
ΔH°f (kJ/mol) : -187.7 -136.3
ΔG°f (kJ/mol) : -120.4 -105.6
S°(J/K·mol) : 109.6 232.7
A) 120°C
B) 144°C
C) 196°C
D) 418°C
E) 585°C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following phase changes decreases the entropy of the system?
A) Melting
B) Heating a gas
C) Vaporization
D) Condensation
E) Sublimation
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In 1774 Joseph Priestley prepared the element oxygen by heating mercury(II) oxide: HgO(s) → Hg(l) + ½O2(g) For this reaction, ΔH° = 90.84 kJ and ΔS° = 108 J/K. Which of the following statements is true?
A) The reaction is only spontaneous at low temperatures.
B) The reaction is spontaneous at all temperatures.
C) ΔG° becomes less favorable as the temperature is raised.
D) The reaction is spontaneous only at high temperatures.
E) The reaction is spontaneous under standard conditions at 25°C.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? HgS(s) + O2(g) → Hg(l) + SO2(g)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following results in a decrease in the entropy of the system?
A) O2(g) , 300 K → O2(g) , 400 K
B) H2O(s) , 0°C → H2O(l) , 0°C
C) N2(g) , 25°C → N2(aq) , 25°C
D) NH3(l) , -34.5°C → NH3(g) , -34.5°C
E) 2H2O2(g) → 2H2O(g) + O2(g)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔG° for the reaction SiCl4(g) + 2Mg(s) → 2MgCl2(s) + Si(s) Substance: SiCl4(g) Mg(s) MgCl2(s) Si(s) ΔG°f (kJ/mol) : -616.98 0 -591.79 0
A) 566.60 kJ
B) 50.38 kJ
C) 25.19 kJ
D) -25.19 kJ
E) -566.60 kJ
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 84
Related Exams