A) testosterone
B) progesterone
C) bile salts
D) estrogen
E) prostaglandins
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following best describes a proton?
A) one negative charge,no mass,found in orbitals
B) no charge,mass of one,found in nucleus
C) one positive charge,mass of one,found in nucleus
D) subatomic particle with no electric charge
E) None of these choices is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Normal pH range for blood is 7.35 to 7.45.If blood pH falls below 7.35,
A) the number of red blood cells decreases.
B) the blood becomes saltier.
C) an imbalance called acidosis results.
D) an imbalance called alkalosis results.
E) nothing happens as this is an acceptable deviation.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
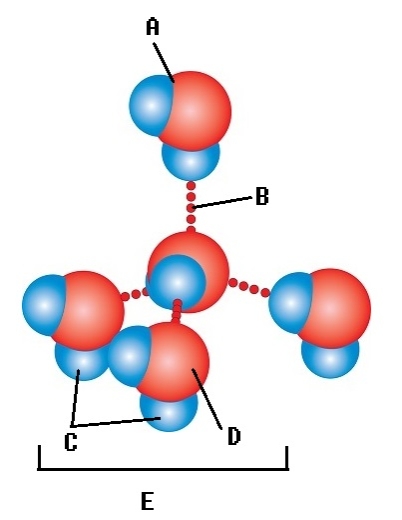 -Water accounts for 50% of the weight of a young adult female and 60% of a young adult male.What kind of molecule is found at "E"?
-Water accounts for 50% of the weight of a young adult female and 60% of a young adult male.What kind of molecule is found at "E"?
A) water molecule
B) polar covalent bond
C) hydrogen atom
D) oxygen atom
E) hydrogen bond
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
ATP
A) serves as the energy currency of the cell.
B) can store,but cannot release energy in the cell.
C) is a sugar found in transfer RNA.
D) stores genetic information.
E) is a nucleotide found in DNA.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom of chlorine has 17 protons and 18 neutrons.Which of the following statements is true?
A) Chlorine has an atomic number of 18.
B) Chlorine atoms have 18 electrons.
C) Chlorine has an atomic number of 35.
D) Chlorine has 35 electrons.
E) Chlorine has a mass number of 35.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molecule used most frequently by cells as a fuel belongs to which of the following groups?
A) carbohydrates
B) nucleic acids
C) prostaglandins
D) phospholipids
E) steroids
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Normal pH for blood is 7.35 to 7.45.Maintenance of the pH in this range is
A) critical because enzymes work best within narrow ranges of pH.
B) not critical because extreme pH values do not affect enzyme function.
C) called denaturation.
D) not required.
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Glycogen is the
A) storage carbohydrate in animals.
B) storage carbohydrate in plants.
C) sugar found in RNA.
D) major nutrient for most body cells.
E) nondigestible plant polysaccharide.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The primary structure of a protein is
A) the hydrogen bonds between amino acids.
B) the folded,helical nature of the molecule.
C) represented by multiple polypeptide chains.
D) the sequence of amino acids in the polypeptide chain.
E) the number of polypeptide chains in the molecule.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following organic groups does a steroid belong to?
A) carbohydrate
B) nucleic acid
C) protein
D) lipid
E) vitamin
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molecular formula H2O means
A) 1 hydrogen atom and 2 oxygen atoms.
B) 1 hydrogen atom and 1 oxygen atom.
C) 2 hydrogen atoms and 1 oxygen atom.
D) 2 hydrogen atoms and 2 oxygen atoms.
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following chemical reactions best represents the decomposition of ATP?
A) ATP + ADP ATP
B) ATP + energy ADP + H2O
C) ATP + H2O ADP + Pi + energy
D) ADP + ADP + ADP ATP
E) ADP + Pi + energy ATP + H2O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The model that helps explain how an enzyme works is the
A) three-dimensional model.
B) denaturation model.
C) activation model.
D) lock-and-key model.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cation is
A) a molecule that conducts electricity when placed in solution.
B) an alteration in the three-dimensional structure of a protein.
C) a positively charged ion.
D) a combination of atoms held together by chemical bonds.
E) a negatively charged ion.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A substance composed of two or more different types of atoms is a(n)
A) element.
B) compound and a molecule.
C) compound.
D) molecule.
E) ion.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following organic groups does an enzyme belong to?
A) nucleic acid
B) lipid
C) vitamin
D) carbohydrate
E) protein
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is false?
A) Water transports nutrients in the body.
B) Water evaporation heats the body.
C) Water evaporation cools the body.
D) Water serves as an effective lubricant in our bodies.
E) Water allows the body to resist sudden temperature changes.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
X-rays can be used to view bones because
A) x-rays can not pass through bone.
B) x-rays pass through bone.
C) x-rays react with bone.
D) bones are less dense than soft tissue.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an x-ray film of the skeletal system,the dense tissue areas appear ______ because they ______ the x-rays; and the less dense tissues appear ______ because they ______ the x-rays.
A) light,absorb; dark,do not absorb
B) dark,absorb; light,do not absorb
C) dark,do not absorb; light,do absorb
D) light,do not absorb; dark,aborb
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 141 - 160 of 168
Related Exams